Acceptance Volume of Expansion and Compression Tanks – Part 3
|
In part 2 of the R L Deppmann Monday Morning Minutes we determined the initial and final temperatures of the hydronic system in order to calculate the expansion tank selection. When we heat the water in a hydronic system, the water expands. This expansion is expressed as Ew in the formula:
Ew – Ep = The expansion of the water minus the expansion of the pipe.
This expansion is calculated for you in the Bell and Gossett ESP-PLUS selection program when you enter the initial and final temperatures and the fluid. The term Ew is not a mystery. It is expressed as:
Ew = [((specific volume at hot Temp.)/ (specific volume at cold Temp.))-1]
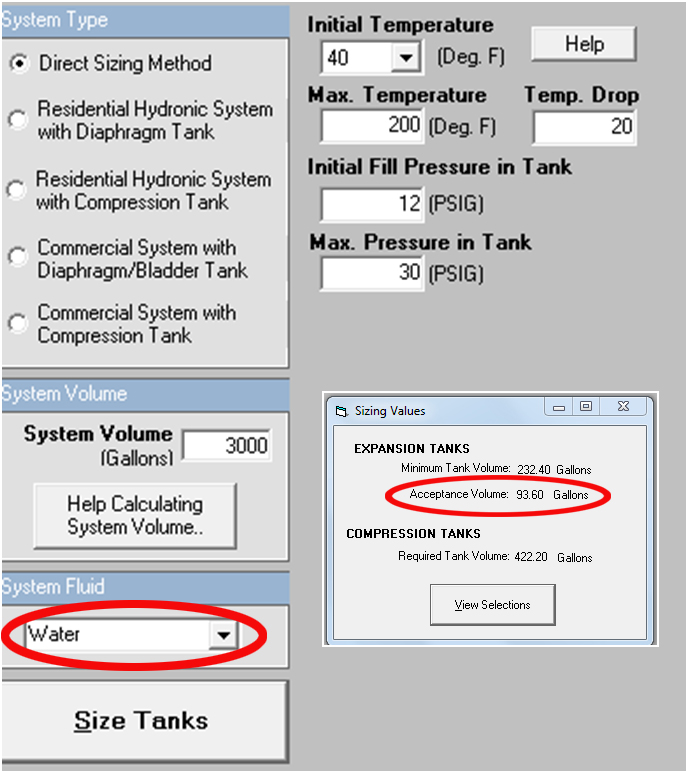
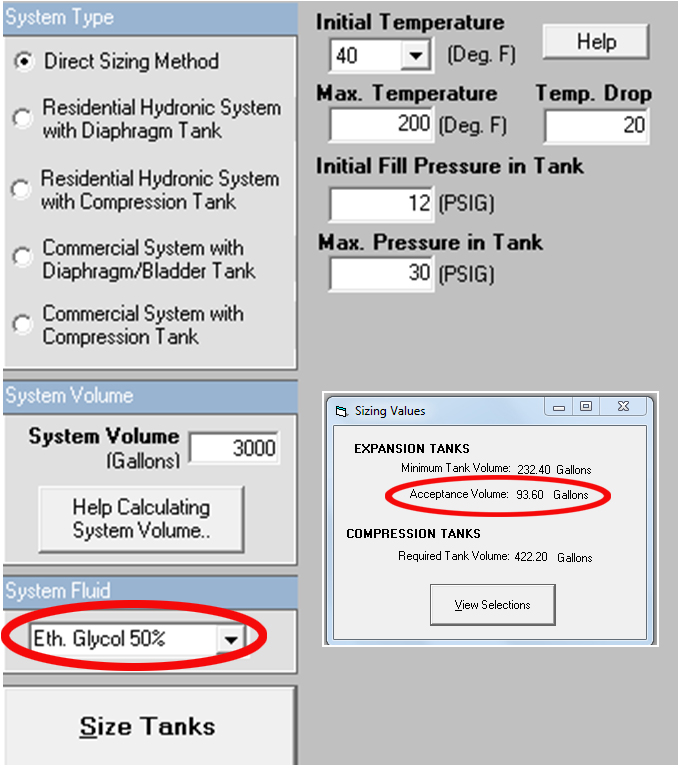
The calculation of the required tank acceptance volume is (Ef – Ep) X system volume. Let’s look at an example and assume the system has 50% Ethylene Glycol but you did not change the default from water.: Assume you have a closed hydronic heating system with a maximum temperature of 200°F and a 20°F ΔT. Assume the volume is 3000 gallons. What happens to the acceptance volume? The acceptance volume required in this example using 50%.
 |
 |
| The acceptance volume for water is 93.6 gallons |
The acceptance volume for 50% E.G. is 125.4 gallons |
As you can see in the example, forgetting to change the fluid type default results in a tank which is 25% undersized. The result of this might be weeping pressure relief valves at the elevated temperatures.
|
Disclaimer: R. L. Deppmann and it’s affiliates can not be held liable for issues caused by use of the information on this page. While the information comes from many years of experience and can be a valuable tool, it may not take into account special circumstances in your system and we therefore can not take responsibility for actions that result from this information. Please feel free to contact us if you do have any questions.
See the rest of this series:




